|
We report here that surface-expressed β protein of GBS binds to human CEACAM1 and CEACAM5 receptors. However, several putative adhesins have no host binding partner characterised. GBS adhesins have been identified to bind extracellular matrix components and cellular receptors. A critical step to infection is adhesion of bacteria to epithelial surfaces. Streptococcus agalactiae, also known as group B Streptococcus (GBS), is the major cause of neonatal sepsis in humans. 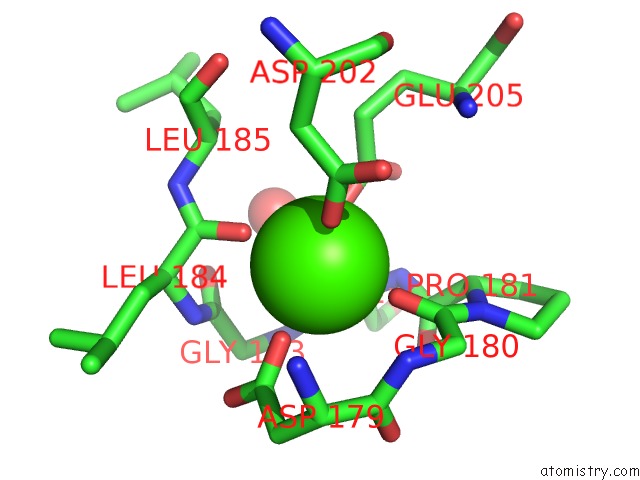
12 Present address: Department of Medical Microbiology, Infection Prevention and Netherlands Reference Laboratory for Bacterial Meningitis, Amsterdam University Medical Center, University of Amsterdam, Amsterdam, The Netherlands.11 Department of Chemistry, Division of Applied Microbiology, Lund University, Lund, Sweden.10 Department of Biochemistry, Emory University School of Medicine, Atlanta, GA, USA.9 Department of Infectious Disease, MRC Centre for Molecular Bacteriology & Infection, Imperial College London, London, UK.8 Department of Laboratory Medicine, Division of Medical Microbiology, Lund University, Lund, Sweden.7 Department of Medical Biosciences, Umeå University, Pathology, Umeå, Sweden.6 Department of Pathology, Sumy State University, Sumy, Ukraine.5 Institute of Anatomy, Medical Faculty, University Duisburg-Essen, Essen, Germany.4 Department of Biochemistry and Biophysics, Science for Life Laboratory, Stockholm University, Stockholm, Sweden.3 Department of Immunology & Microbiology, University of Colorado Anschutz Medical Campus, Aurora, CO, USA.2 Institute of Human Virology, University of Maryland School of Medicine, University of Maryland, Baltimore, MD, USA.1 Department of Medical Microbiology, University Medical Center Utrecht, Utrecht University, Utrecht, The Netherlands.A list of Web resources in the area of membrane protein crystallogenesis is included. The three new methods are rationalized by analogy to crystallization in microgravity and with respect to epitaxy. The experimental details of both methods are reviewed and their general applicability in the future is commented on. Thus far, the bicelle and vesicle-fusion methods have produced crystals of one membrane protein, bacteriorhodopsin. The section ends with a hypothesis for nucleation and growth of membrane protein crystals in meso. Practical aspects of the method are examined with regard to salt, detergent, and screen solution effects crystallization at low temperatures tailoring the cubic phase to suit the target protein different cubic-phase types dealing with low-protein samples, colorless proteins, microcrystals, and radiation damage transport within the cubic phase for drug design, cofactor retention, and phasing using spectroscopy for quality control harvesting crystals and miniaturization and robotization for high-throughput screening. The cubic-phase method is described from the following perspectives: how it is done in practice, its general applicability and successes to date, and the nature of the mesophases integral to the process. The experimental challenges associated with and the solutions for procuring adequate amounts of homogeneous membrane proteins, or parts thereof, are examined. In the belief that a knowledge of the underlying phase science is integral to understanding the molecular basis of these assorted crystallization strategies, the article begins with a brief primer on lipids, mesophases, and phase science, and the related issue of form and function as applied to lipids is addressed. 
The focus instead is on the latest methods, all of which exploit the spontaneous self-assembling properties of lipids and detergent as vesicles (vesicle-fusion method), discoidal micelles (bicelle method), and liquid crystals or mesophases (in meso or cubic-phase method). The long-established surfactant-based method has been reviewed extensively and is not examined in detail here. An overview of the current ideas and experimental approaches prevailing in the area of membrane protein crystallization is presented. The rate-limiting step from protein to structure is crystal production.  
Currently, the only reliable way to get it is crystallographically. The need for high-resolution structure information on membrane proteins is immediate and growing.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
